Essential cancer medicines
Essential medicines are defined as those that “satisfy the priority health care needs of the population”.

Cancer is the second leading cause of death globally. Ensuring the availability of medicines for treatment – including chemotherapy, immunotherapy, antibiotics and for pain relief – is crucial. Many countries around the world, however, struggle with issues of access, availability and affordability of quality-assured essential medicines for cancer treatment.
Essential medicines are defined as those that “satisfy the priority health care needs of the population”. They are selected with regard to their public health relevance, evidence on efficacy and safety, and comparative cost-effectiveness. They are intended "to be available within the context of functioning health systems at all times in adequate amounts, in the appropriate dosage forms, with assured quality and adequate information, and at a price the individual and the community can afford[1].” (The Selection of Essential Medicines, WHO)
In 1975 the World Health Assembly requested WHO to create a guidance tool that would help member states in selecting and procuring quality-assured essential medicines. The first WHO Model List of Essential Medicines list (WHO EML) was published two years later, establishing a guiding principle that some medicines were more useful than others and that these medicines were often not available to all who needed them.[2] Selection of these medicines was initially opinion-based, relying on a physician’s experience in treating patients; now the list is based on an approach grounded in scientific evidence.
In 2015, UICC convened experts to review the cancer medicines listed on the WHO EML. As a result, the methodology for the selection of essential cancer medicines was revised to propose medicines based on specific regimens for specific cancers, which would make it easier for policymakers to identify cancer medicines relevant for their national disease burden.[3] Using this process, the WHO EML was further updated, with the addition of cancer medicines in 2017 and 2019. In the EML review for 2021, more than a quarter of applications were for cancer medicines, showing the pressing need for cancer medicines to be available for populations globally in light of the rising burden of cancer.
Improving access to essential cancer medicines

Source: Bull World Health Organ 2019;97:394-404C doi: http://dx.doi.org/10.2471/BLT.18.222448
It is particularly important that these essential medicines are available for cancer patients in a timely manner, as the likelihood of a better outcome and quality of life increases when the cancer is detected and treated early.
Yet, many countries – particularly in low-resource settings – struggle with numerous barriers to access. These barriers include the pricing and quality of medicines, procurement and supply chain issues, diagnostic capacity, infrastructure and the lack of adequate human resources.
As of 2019, comprehensive cancer services were only reported to exist in fewer than 15% of low- and middle-income countries, against 90% of high-income countries. Less than 50% of cancer medicines on the WHO EML are still not available to patients living in LMICs and more than 80% of the world’s population has little or no access to pain medicines, such as morphine and other opioids.
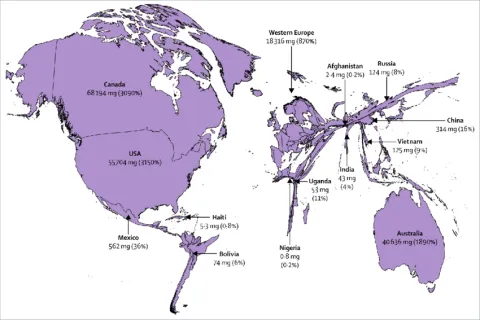
Distributed opioid morphine-equivalent (morphine in mg/patient in need of palliative care, average 2010–13), and estimated percentage of need that is met for the health conditions most associated with serious health-related suffering
Source: Knaul FM, Farmer PE, Krakauer El, et al (2017).
The incredible progress made in making other medicines more accessible, such as antiviral medicines for HIV/AIDS and antiviral medicines for hepatitis C, shows that this situation can be changed. To do so requires strong government commitment, and a multi-stakeholder approach to strengthen health systems and build effective and sustainable procurement and supply chain processes to improve the delivery of cancer medicines so the appropriate treatment reaches the right patients at the right time.
Establishing a comprehensive National List of Essential Medicines (NEML) tailored to the needs of the population is a key first step for a country to improve the accessibility and availability of cancer medicines. Currently, there is a notable discrepancy in many countries between their NEML and the WHO EML: many essential cancer medicines, are simply not included on the NEMLs.
Another crucial step is to support countries in developing sustainable health financing and the capacity for the appropriate diagnosis of cancer, as well as the proper handling and supply monitoring of cancer medicines. In this regard, it is essential to address regulatory capacity, procurement, forecasting and supply chain management.
Addressing the issue of affordability is another issue that needs to be addressed, including for those medicines still on patent. Various approaches can come into play here including the use of public-health oriented voluntary licences for essential medicines and by supporting manufacturers of generic and biosimilar medicines in developing, registering and supplying quality-assured medicines at affordable prices.
National Lists of Essential Medicines (NEML)
The selection of essential medicines is one of the core components of a national medicines policy. The WHO EML serves as a model to help countries prioritise what is included in their NEMLs for public procurement.
The selection of essential medicines is one of the core components of a national medicines policy. The WHO EML serves as a model to help countries prioritise what is included in their NEMLs for public procurement.
A NEML can decrease barriers to access by maintaining focus on a limited list of medicines. However, lists are not always updated in a timely manner. An analysis of 135 NEMLs in 2016, found that of the 25 cancer medicines on the 2013 WHO EML and 16 added in 2015, a median of 17 and 3 appeared in NEMLs respectively.[4]
The addition of a medicine on the NEML is a crucial step towards ensuring its availability. These lists should be updated at regular intervals.
View the step by step process countries can use in revising their NEMLs. →
The role of UICC and its members
Establishing a NEML is crucial; however, additional steps must be taken to overcome barriers and ensure access to medicines.
These challenges are increasingly being raised in discussions between policymakers, civil society and the pharmaceutical industry, as governments move towards Universal Health Coverage.
Building on this momentum, UICC was instrumental in bringing the cancer community together for an open, inclusive, constructive, and evidence-based dialogue about how to pursue solutions collectively to improve access to cancer medicines, for the wellbeing of patients and sustainability of cancer care and health systems.
Members can pursue this issue and encourage governments and policymakers to ensure the implementation of an evidence-based NEML, in alignment with the WHO Model List of Essential Medicines, as a first step in improving access to cancer medicines.


The ATOM Coalition: Strengthening access to essential oncology medicines in low- and lower-middle income countries
Launched in May 2022, the Access to Oncology Medicines (ATOM) Coalition is a global partnership led by the Union for International Cancer Control (UICC). The Coalition of more than 40 leading organisations from civil society, and the public and private sectors, works collectively to address the access barriers to essential cancer medicines in low- and lower middle-income countries (LLMICs).
Increasing access to WHO EML cancer medicines
The ATOM Coalition’s Medicines Expert Advisory Group was established to identify and prioritise the essential cancer medicines that the ATOM Coalition will initially focus on.
An international team of experts leads the Advisory Group, including members of the WHO Expert Committee on Selection and Use of Essential Medicines, members from the Medicines Patent Pool Advisory Committee, as well as representatives from Access to Medicine Foundation and the American Society for Clinical Pathology. A key recommendation by the group, which has been adopted by the ATOM Coalition, is to prioritise medicines listed on the WHO Model List of Essential Medicine that target those cancers causing the heaviest mortality in LLMICs: lung, colorectal, breast, cervical, prostate and childhood cancers.
The priority list consists of two parts. The first is a list of patented medicines for cancer currently listed on the WHO EML or likely to be included in future revisions. The expert advisory group provides guidance to the Coalition on which medicines from this list to pursue voluntary licenses facilitated by Medicines Patented Pool.
Additionally, the Advisory Group has developed a second priority list of approximately 17 generic and biosimilar medicines to focus on, with an initial focus on breast cancer, before expanding focus to other high-priority cancers mentioned above. The Access to Medicine Foundation’s new industry programme on generic manufacturers and their work on a product list for cancer medicines helped to inform this second list.
In its first phase, the ATOM Coalition will focus its efforts on improving access to these prioritised medicines in the 46 countries targeted by the ATOM Coalition. In the coming year, the list is expected to expand to cover more medicines and cancers.
The Access to Oncology Medicines (ATOM) Coalition ambition's is "to reduce suffering and deaths caused by cancer in low- and lower-middle income countries through improved access to and use of essential cancer medicines".
Useful links for further reading
References
- [1] http://www.who.int/medicines/areas/access/NPrices_Glossary.pdf
- [2] 25 years of the WHO essential medicines lists: progress and challenges https://apps.who.int/medicinedocs/documents/s17543en/s17543en.pdf
- [3] Shulman LN, Wagner CM, Barr R, et al. Proposing essential medicines to treat cancer: methodologies, processes, and outcomes. J Clin Oncol. 2016; 34:69–75.
- [4] Robertson J, Barr R, Shulman LN, Forte GB, Magrini N. Essential medicines for cancer: WHO recommendations and national priorities. Bulletin World Health Organization. 2016;94:735–742.
Last update
Monday 30 March 2026